Manufacturing Unit
The production facility is state-of-art with modem dedicated equipments and latest technology with absolute compliance to, current Good Manufacturing Practice (cGMP). The entire process of WFI production takes place in completely sterilized environment. Filling, packing etc. are fully automatic, untouched by human hand which eliminates any risk of contamination.
PYROGEN FREE PURIFIED WATER
At our plant, Purified Pyrogen Free WFI is being produced using a combination of Chlorination, Sand Filter, Softner , Ultra Filtration (UF) Millipore, Double Pass Reverse Osmosis(RO), Continuous Electro-Deionisation (CEDI) and UV Light treatment which helps to remove most particle, pyrogens, heavy minerals, colloids, microorganisms and also kills microbes.Further, this purified water undergoes Multi-Column Distillation removing Endotoxins. Finally WFI is filtered through 0.2u Filter.Produces high grade purified water.
This Pyrogen Free, Purified Water is stroed in Stroage Tanks at above 85°C.
- Double-pass Reverse Osmosis (RO) system with UV and hot water sanitization ensures NIL bacteria.
- EDI (Electro-De-lonizing) to ensure perfect CONDUCTIVITY.
- Multl-column Distillation plant based on FINN AQUA design with Pure Steam Generator (PSG).and Degasser, which ensures to produce pure, particle and pyrogen-free, water for injection as per IP/BP/USP Specifications.
- Continuous on-line TOC Analyzer counts for perfect carbon free water,
- Suitable CIP/SIP programs for automatic cleaning, sanitization and sterilization.
- All contact parts are made out of AISI 316 L qualities S.S and are electro mirror polished.
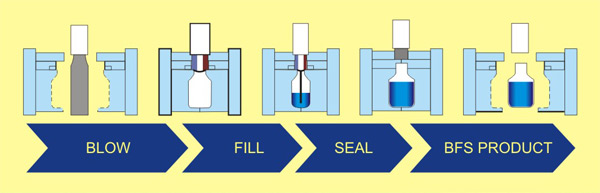
3-IN-1 ASEPTIC "BLOW FILL SEAL" (BFS) TECHNOLOGY
BLOW FILL SEAL (BFS) system is an advanced aseptic technology which combines many processes into one machine. It extrudes pharma grade PP/PE material into moulds, Blow, Fill and Seal functions fully automatically under Aseptic conditions. High risk Filling-sealing is performed under Class A/100 Area Air flushing device which helps to protect from outer air pollution BLOW FILL SEAL (BFS) with back ground of class 10,000 with BFS technology machine fitted with SIP and CIP system for cleaning with digital monitoring PLC.
QUALITY MANAGEMENT SYSTEMS
Quality Management System aims -at providing world class quality standard with its highly experienced and qualified personnel having expertise in various disciplines like Chemistry, Microbiology, Pharmacy ‘and other relevant areas. The process of Quality Control and Quality Assurance starts right from ‘the stage of procurement of raw material to delivery of finished product.
The Quality control laboratory is well equipped, having separate area for Chemistry, Instrument and Microbiology.For regular analysis, pH meter, Conductivity meter, ultrasonic bath, Digital muffle furnace, Hotplate with magnetic stirrer, Vortex mixer, Digital colony counter, Microscope, BOD incubator, Bacteriological incubator, Hot air oven, Analytical Balance, LAF units, Endotoxin detection and Sterility testing facility is provided.
THE PRODUCED STERILE WFI CONTAINS NO ADDEO SUBSTANCES IT MEETS THE REQUIREMENT FOR CHEMICAL (CONDUCTIVITY, TOC, PH, NITRATES AND HEAVY METALS ETC.) AND MICROBIOLOGICAL REQUIREMENTS (BIO-BURDEN AND ENDO TOXINS).
SUNLIFE SWFI MATCHES USP_SPECIFICATIONS i.e. pH: 5.O to 7.0, CONDUCTIVITY: 1.3 µs/cm, TOC: 500 ppb, BACTERIAL ENDOTOXIN: 0.25 IU/ml.SunLife.Sterile Water for injection (SWFI) is the Sterile Water For Injection: SWFI is a sterile, non-pyrogenic preparation which contains no bacteriostat, antimicrobial agent or added buffer and is supplied only in single dose Plastic containers with easy "twist-off" seal; to dilute or' dissolve drugs for injection and reconstitution of dry powder'.We welcome our business partners and associates to visit our facility to have confidence on the quality, capacity and capabilities.